Online first
About the Journal
Current issue
Archive
Publication Ethics
Anti-Plagiarism system
Instructions for Authors
Instructions for Reviewers
Editorial Office
Editorial Board
Contact
Reviewers
All Reviewers
2025
2024
2023
2022
2021
2020
2019
2018
2017
2016
General Data Protection Regulation (RODO)
CASE REPORT
Isobolographic analysis of interactions – a pre-clinical perspective
1
Department of Occupational Medicine, Medical University, Lublin, Poland
2
Department of Diagnostics and Microsurgery of Glaucoma, Medical University, Lublin, Poland
3
Department of Experimental Pharmacology, Institute of Rural Health, Lublin, Poland
Corresponding author
J Pre Clin Clin Res. 2023;17(4):238-241
KEYWORDS
TOPICS
ABSTRACT
Introduction:
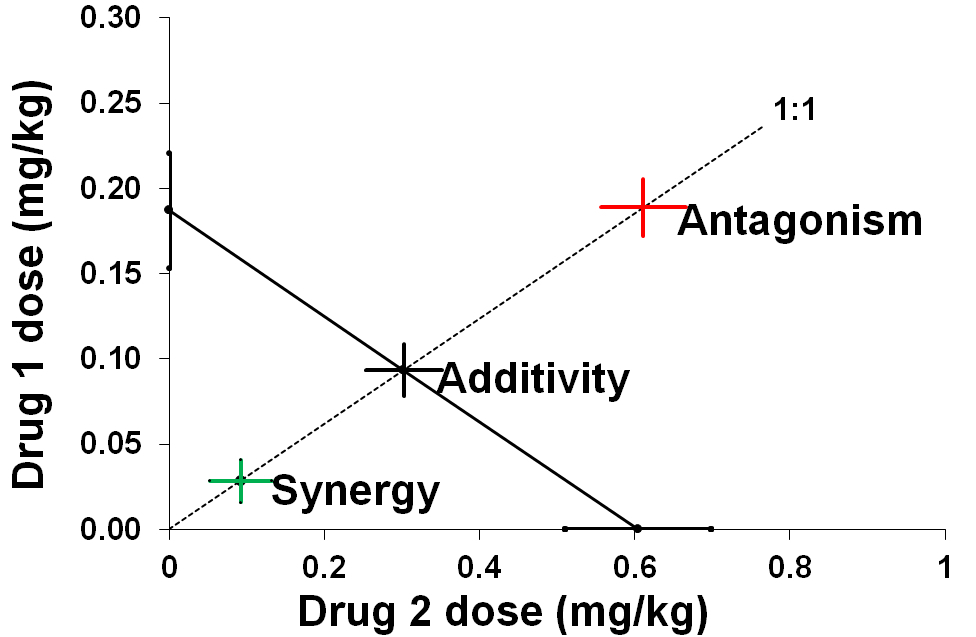
Isobolographic analysis is the preferred method of assessment of pharmacodynamic interactions occurring among drugs administered in mixture in both pre-clinical and clinical studies. Despite its mathematical complexity, rigorous preliminary conditions and various prerequisites to be met, it assesses the pharmacodynamic interactions, classifying them as additive, antagonistic, synergistic or indifferent in nature. These interactions are usually plotted in the Cartesian system of coordinates forming isobolograms. The strength (power) of interactions is calculated and presented as an interaction index.
Conclusions:
This report provides basic information on the isobolographic analysis used experimentally in preclinical conditions indicating the underestimation of this valuable method in pharma
Isobolographic analysis is the preferred method of assessment of pharmacodynamic interactions occurring among drugs administered in mixture in both pre-clinical and clinical studies. Despite its mathematical complexity, rigorous preliminary conditions and various prerequisites to be met, it assesses the pharmacodynamic interactions, classifying them as additive, antagonistic, synergistic or indifferent in nature. These interactions are usually plotted in the Cartesian system of coordinates forming isobolograms. The strength (power) of interactions is calculated and presented as an interaction index.
Conclusions:
This report provides basic information on the isobolographic analysis used experimentally in preclinical conditions indicating the underestimation of this valuable method in pharma
Łuszczki JJ, Wlaź A. Isobolographic analysis of interactions: a pre-clinical perspective. J Pre-Clin Clin Res. 2023; 17(4): 238–241. doi: 10.26444/jpccr/177246
REFERENCES (39)
1.
Tallarida RJ. Interactions between drugs and occupied receptors. Pharmacol Ther. 2007;113(1):197–209. https://doi.or /10.1016/j. pharmthera.2006.08.002.
2.
Dawson DA, Allen EM, Allen JL, et al. Time-dependence in mixture toxicity prediction. Toxicology. 2014;326:153–163. https://doi. org/10.1016/j.tox.2014.10.015.
3.
Świeczkowski D, Zdanowski S, Merks P, et al. The plague of unexpected drug recalls and the pandemic of falsified medications in cardiovascular medicine as a threat to patient safety and global public health: A brief review. J Cardiol. 2022;29(1):133–139. https://doi.org/10.5603/ CJ.a2020.0168.
4.
Chou TC, Motzer RJ, Tong Y, Bosl GJ. Computerized quantitation of synergism and antagonism of taxol, topotecan, and cisplatin against human teratocarcinoma cell growth: a rational approach to clinical protocol design. J Nat Cancer Inst. 1994;86(20):1517–1524. https://doi. org/10.1093/jnci/86.20.1517.
5.
Chou TC, Talalay P. Quantitative analysis of dose-effect relationships: the combined effects of multiple drugs or enzyme inhibitors. Adv Enzym Regul. 1984;22:27–55. https://doi.org/10.1016/0065-2....
6.
Chou TC. Theoretical basis, experimental design, and computerized simulation of synergism and antagonism in drug combination studies. Pharmacol Rev. 2006;58(3):621–681. https://doi.org/10.1124/pr.58.....
7.
Tallarida RJ. Revisiting the isobole and related quantitative methods for assessing drug synergism. J Pharmacol Exp Ther. 2012;342(1):2–8. https://doi.org/10.1124/jpet.1....
8.
Tallarida RJ, Stone DJ, Raffa RB. Efficient designs for studying synergistic drug combinations. Life Sci. 1997;61(26):Pl 417–425. https:// doi.org/10.1016/s0024-3205(97)01030-8.
9.
Gessner PK. Isobolographic analysis of interactions: an update on applications and utility. Toxicology. 1995;105(2–3):161–179. https://doi.org/10.1016/0300-4....
10.
Tallarida RJ. Quantitative methods for assessing drug synergism. Genes Cancer 2011;2(11):1003–1008. https://doi.org/10.1177/194760....
11.
Tallarida RJ, Porreca F, Cowan A. Statistical analysis of drug-drug and site-site interactions with isobolograms. Life Sci. 1989;45(11):947–961. https://doi.org/10.1016/0024-3....
12.
Poch G, Dittrich P, Holzmann S. Evaluation of combined effects indose-response studies by statistical comparison with additive and independent interactions. J Pharmacol Methods. 1990;24(4):311–325. https://doi.org/10.1016/0160-5....
13.
Poch G, Dittrich P, Reiffenstein RJ, Lenk W, Schuster A. Evaluation of experimental combined toxicity by use of dose-frequency curves: comparison with theoretical additivity as well as independence. Can J Physiol Pharmacol. 1990;68(10):1338–1345. https://doi.org/10.1139/y90-20....
14.
Poch G, Reiffenstein RJ, Unkelbach HD. Application of the isobologram technique for the analysis of combined effects with respect to additivity as well as independence. Can J Physiol Pharmacol. 1990;68(6):682–688. https://doi.org/10.1139/y90-10....
16.
Tallarida RJ. An overview of drug combination analysis with isobolograms. J Pharmacol Exp Ther. 2006;319(1):1–7. https://doi. org/10.1124/jpet.106.104117.
17.
Tallarida RJ. Drug combinations: tests and analysis with isoboles. Curr Protoc Pharmacol. 2016;72:9.19.1–19. https://doi.org/10.1002/047114....
18.
Wawruszak A, Okon E, Telejko I, Czerwonka A, Łuszczki J. Additive pharmacological interaction between sirtuin inhibitor cambinol and paclitaxel in MCF7 luminal and MDA-MB-231 triple-negative breast cancer cells. Pharmacol Rep. 2022;74(5):1011–1024. https://doi. org/10.1007/s43440-022-00393-w.
19.
Wawruszak A, Łuszczki J, Okon E, et al. Antagonistic Pharmacological Interaction between Sirtuin Inhibitor Cambinol and Paclitaxel in Triple- Negative Breast Cancer Cell Lines: An Isobolographic Analysis. Int J Mol Sci. 2022;23(12):6458. https://doi.org/10.3390/ijms23....
20.
Gumbarewicz E, Łuszczki JJ, Wawruszak A, et al. Isobolographic analysis demonstrates additive effect of cisplatin and HDIs combined treatment augmenting their anti-cancer activity in lung cancer cell lines. Am J Cancer Res. 2016;6(12):2831–2845.
21.
Wawruszak A, Łuszczki JJ, Grabarska A, et al. Assessment of interactions between cisplatin and two histone deacetylase inhibitors in MCF7, T47D and MDA-MB-231 human breast cancer cell lines – an isobolographic analysis. PloS One 2015;10(11):e0143013. https://doi.org/10.1371/journa.... pone.0143013.
22.
Łuszczki JJ. Isobolographic analysis of interaction for three-drug combination of carbamazepine, phenobarbital and topiramate in the mouse maximal electroshock-induced seizure model. Pharmacology 2016;97(5–6):259–264. https://doi.org/10.1159/000444....
23.
Łuszczki JJ, Andres MM, Czuczwar SJ. Synergistic interaction of gabapentin and oxcarbazepine in the mouse maximal electroshock seizure model--an isobolographic analysis. Eur J Pharmacol. 2005;515(1–3):54–61. https://doi.org/10.101 /j.ejphar.2005.03.046.
24.
Łuszczki JJ, Andres-Mach MM, Ratnaraj N, Patsalos PN, Czuczwar SJ. Levetiracetam and felbamate interact both pharmacodynamically and pharmacokinetically: an isobolographic analysis in the mouse maximal electroshock model. Epilepsia 2007;48(4):806–815. https://doi.org/10.1111/j.1528....
25.
Łuszczki JJ, Antkiewicz-Michaluk L, Raszewski G, Czuczwar SJ. Interactions of 1-methyl-1,2,3,4-tetrahydroisoquinoline with lamotrigine, oxcarbazepine, pregabalin, and topiramate in the mouse maximal electroshock-induced seizure model: a type I isobolographic analysis. Epilepsy Res. 2010;89(2–3):207–219. https://doi.org/10.1016/j. eplepsyres.2010.01.001.
26.
Zadrożniak M, Szymański M, Łuszczki JJ. Vitamin C alleviates ototoxic effect caused by coadministration of amikacin and furosemide. Pharmacol Rep. 2019;71(2):351–356. https://doi.org/10.1016/j. pharep.2019.01.002.
27.
Zadrożniak M, Szymański M, Łuszczki JJ. N-Acetyl-L-cysteine Affects Ototoxicity Evoked by Amikacin and Furosemide Either Alone or in Combination in a Mouse Model of Hearing Threshold Decrease. Int J Mol Sci. 2023;24(8):7596. https://doi.org/10.3390/ijms24....
28.
Isiordia-Espinoza MA, Terán-Rosales F, Serafín-Higuera NA, et al. Isobolographic analysis of the ciprofloxacin-gentamicin combination against beta-lactamase-producing Staphylococcus aureus. Fund Clin Pharmacol. 2023;37(6):1198–1204. https://doi.org/10.1111/fcp.12....
29.
Gómez-Sánchez E, Franco-de la Torre L, Hernández-Gómez A, et al. Antagonistic, synergistic, and additive antibacterial interaction between ciprofloxacin and amoxicillin against Staphylococcus aureus. Fundam Clin Pharmacol. 2023;37(1):174–181. https://doi.org/10.1111/fcp.12....
30.
Kissin I. Antinociceptive Agents as General Anesthetic Adjuncts: Supra-additive and Infra-additive Interactions. Anesthesia Analgesia 2023;137(6):1198–1207. https://doi.org/10.1213/ANE.00....
31.
Rojas-Aguilar FA, Briones-Aranda A, Jaramillo-Morales OA, et al. The Additive Antinociceptive Effect of Resveratrol and Ketorolac in the Formalin Test in Mice. Pharmaceuticals 2023;16(8):1078. https://doi. org/10.3390/ph16081078.
32.
Łuszczki JJ, Kolacz A, Wojda E, et al. Synergistic interaction of gabapentin with tiagabine in the hot-plate test in mice: an isobolographic analysis. Pharmacol Rep. 2009;61(3):459–467. https://doi.org/10.1016/ s1734-1140(09)70087-8.
33.
Łuszczki JJ, Pałka J, Marzęda P, et al. Antinociceptive screening of various 1,2,4-triazole-3-thione derivatives in the hot-plate test in mice. J Pre-Clin Clin Res. 2019;13(1):9–12. https://doi.org/10.26444/ jpccr/105514.
34.
Dai G, Li B, Xu Y, et al. Synergistic interaction between matrine and paracetamol in the acetic acid writhing test in mice. Eur J Pharmacol. 2021;895:173869. https://doi.org/10.1016/j.ejph....
35.
Leksiri S, Hasriadi, Dasuni Wasana PW, et al. Co-administration of Pregabalin and Curcumin Synergistically Decreases Pain-Like Behaviors in Acute Nociceptive Pain Murine Models. Molecules 2020;25(18):4172. https://doi.org/10.3390/molecu....
36.
Verrelle P, Gestraud P, Poyer F, et al. Integrated High-Throughput Screening and Large-Scale Isobolographic Analysis to Accelerate the Discovery of Radiosensitizers With Greater Selectivity for Cancer Cells. Int J Radiat Oncol Biol Phys. 2023;S0360-3016(23)07958-0. https://doi.org/10.1016/j.ijro....
37.
Gennings C. Economical designs for detecting and characterizing departure from additivity in mixtures of many chemicals. Food Chem Toxicol. 1996;34(11–12):1053–1058. https://doi.org/10.1016/s0278- 6915(97)00074-4.
38.
Poch G, Pancheva SN. Calculating slope and ED50 of additive dose-response curves, and application of these tabulated parameter values. J Pharmacol Toxicol Meth. 1995;33(3):137–145. https://doi.org/10.1016/1056-8....
39.
Tallarida RJ. The interaction index: a measure of drug synergism. Pain 2002;98(1–2):163–168. https://doi.org/10.1016/s0304-....
Share
RELATED ARTICLE
We process personal data collected when visiting the website. The function of obtaining information about users and their behavior is carried out by voluntarily entered information in forms and saving cookies in end devices. Data, including cookies, are used to provide services, improve the user experience and to analyze the traffic in accordance with the Privacy policy. Data are also collected and processed by Google Analytics tool (more).
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.