Online first
About the Journal
Current issue
Archive
Publication Ethics
Anti-Plagiarism system
Instructions for Authors
Instructions for Reviewers
Editorial Office
Editorial Board
Contact
Reviewers
All Reviewers
2025
2024
2023
2022
2021
2020
2019
2018
2017
2016
General Data Protection Regulation (RODO)
RESEARCH PAPER
Rat stromal vascular fraction (rSVF) cells can be directly transduced to express rat TNF-related apoptosis inducing ligand (rTRAIL), and exert similar but less prominent effects compared to rat adipose derived stem cells (rADSC) – an in vitro study
1
Department of Methodology, Medical University of Warsaw, Warsaw, Poland
2
Doctoral School, Medical University of Warsaw, Warsaw, Poland
3
Department of Histology and Embryology, Medical University of Warsaw, Warsaw, Poland
Corresponding author
Wiktor Paskal
Department of Methodology, Medical University of Warsaw, Banacha 1b, 02-091 Warsaw, Poland
Department of Methodology, Medical University of Warsaw, Banacha 1b, 02-091 Warsaw, Poland
KEYWORDS
TOPICS
ABSTRACT
Introduction and objective:
Breast cancer remains a major clinical challenge, with high recurrence rates and the need for safer, more targeted local therapies. Biologic brachytherapy using genetically modified mesenchymal cells offers a promising strategy for localized cytokine delivery. While adipose-derived stem cells (rADSC) have shown potential as carriers of rat TRAIL (rTRAIL), their clinical translation is limited by the need for ex vivo expansion and concerns over tumour-promoting effects.
Material and methods:
The study investigated whether freshly isolated rat stromal vascular fraction (rSVF) cells could be directly transduced with a lentiviral vector to express rTRAIL. Their anticancer activity was compared to rADSC in vitro using co-culture with rat mammary gland cancer cells. The expression was also compared of several markers (CD90, CD44, CD29, CD105, VEGF2, CD31, CD45) between rSVF and rADSC.
Results:
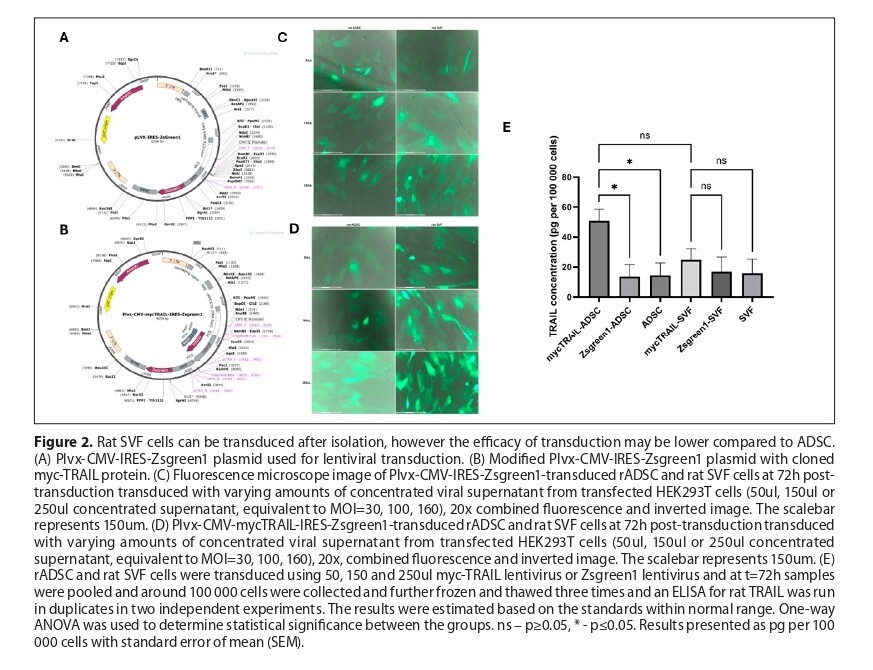
There were no significant differences in the expression of markers between rSVF and rADSC. Both cell types successfully expressed rTRAIL, with rTRAIL-rSVF producing approximately 50% of the TRAIL protein levels observed in rTRAIL-rADSC. In functional assays, rSVF-derived supernatants and co-cultures reduced breast cancer cell viability and colony formation, but the inhibitory effect was approximately 30–50% lower than that of rADSC, depending on the assay and cancer cell line used.
Conclusions:
These findings confirm that rSVF can serve as a transducible and partially effective alternative for rTRAIL delivery and may be valuable for rapid, autologous therapies where culture expansion is not feasible. Further optimization and in vitro validation are needed.
Breast cancer remains a major clinical challenge, with high recurrence rates and the need for safer, more targeted local therapies. Biologic brachytherapy using genetically modified mesenchymal cells offers a promising strategy for localized cytokine delivery. While adipose-derived stem cells (rADSC) have shown potential as carriers of rat TRAIL (rTRAIL), their clinical translation is limited by the need for ex vivo expansion and concerns over tumour-promoting effects.
Material and methods:
The study investigated whether freshly isolated rat stromal vascular fraction (rSVF) cells could be directly transduced with a lentiviral vector to express rTRAIL. Their anticancer activity was compared to rADSC in vitro using co-culture with rat mammary gland cancer cells. The expression was also compared of several markers (CD90, CD44, CD29, CD105, VEGF2, CD31, CD45) between rSVF and rADSC.
Results:
There were no significant differences in the expression of markers between rSVF and rADSC. Both cell types successfully expressed rTRAIL, with rTRAIL-rSVF producing approximately 50% of the TRAIL protein levels observed in rTRAIL-rADSC. In functional assays, rSVF-derived supernatants and co-cultures reduced breast cancer cell viability and colony formation, but the inhibitory effect was approximately 30–50% lower than that of rADSC, depending on the assay and cancer cell line used.
Conclusions:
These findings confirm that rSVF can serve as a transducible and partially effective alternative for rTRAIL delivery and may be valuable for rapid, autologous therapies where culture expansion is not feasible. Further optimization and in vitro validation are needed.
REFERENCES (30)
1.
Gentile D, Martorana F, Karakatsanis A, et al. Predictors of mastectomy in breast cancer patients with complete remission of primary tumor after neoadjuvant therapy: A retrospective study. Eur J Surg Oncol. 2024;50(12):108732. https://doi.org/10.1016/j.ejso....
2.
Pascal W, Gotowiec M, Smoliński A, et al. Biologic Brachytherapy: Genetically Modified Surgical Flap as a Therapeutic Tool—A Systematic Review of Animal Studies. Int J Mol Sci. 2024;25(19):10330. https://doi.org/10.3390/ijms25....
3.
Montinaro A, Walczak H. Harnessing TRAIL-induced cell death for cancer therapy: a long walk with thrilling discoveries. Cell Death Differ. 2023;30(2):237–249. https://doi.org/10.1038/s41418....
4.
Soto-Gamez A, Wang Y, Zhou X, et al. Enhanced extrinsic apoptosis of therapy-induced senescent cancer cells using a death receptor 5 (DR5) selective agonist. Cancer Letters. 2022;525:67–75. https://doi.org/10.1016/j.canl....
5.
Zhang Y, Zhang B. TRAIL Resistance of Breast Cancer Cells Is Associated with Constitutive Endocytosis of Death Receptors 4 and 5. Mol Cancer Res. 2008;6(12):1861–1871. https://doi.org/10.1158/1541-7....
6.
Humphreys LM, Fox JP, Higgins CA, et al. A revised model of TRAIL-R2 DISC assembly explains how FLIP(L) can inhibit or promote apoptosis. EMBO Rep. 2020;21(3):e49254. https://doi.org/10.15252/embr.....
7.
Sarif Z, Tolksdorf B, Fechner H, et al. Mcl-1 targeting strategies unlock the proapoptotic potential of TRAIL in melanoma cells. Mol Carcinog. 2020;59(11):1256–1268. https://doi.org/10.1002/mc.232....
8.
Karimi-Shahri M, Javid H, Sharbaf Mashhad A, et al. Mesenchymal stem cells in cancer therapy; the art of harnessing a foe to a friend. Iran J Basic Med Sci. 2021;24(10):1307–1323. https://doi.org/10.22038/ijbms....
9.
Gil-Chinchilla JI, Zapata AG, Moraleda JM, et al. Bioengineered Mesenchymal Stem/Stromal Cells in Anti-Cancer Therapy: Current Trends and Future Prospects. Biomolecules. 2024;14(7):734. https://doi.org/10.3390/biom14....
10.
Fakiruddin KS, Ghazalli N, Lim MN, et al. Mesenchymal Stem Cell Expressing TRAIL as Targeted Therapy against Sensitised Tumour. Int J Mol Sci. 2018;19(8). https://doi.org/10.3390/ijms19....
11.
Loebinger MR, Eddaoudi A, Davies D, et al. Mesenchymal stem cell delivery of TRAIL can eliminate metastatic cancer. Cancer Res. 2009;69(10):4134–4142. https://doi.org/10.1158/0008-5....
12.
Liu Z, Li S, Ma T, et al. Secreted TRAIL gene-modified adipose-derived stem cells exhibited potent tumor-suppressive effect in hepatocellular carcinoma cells. Immun Inflamm Dis. 2021;9(1):144–156. https://doi.org/10.1002/iid3.3....
13.
Grisendi G, Bussolari R, Cafarelli L, et al. Adipose-Derived Mesenchymal Stem Cells as Stable Source of Tumor Necrosis Factor–Related Apoptosis-Inducing Ligand Delivery for Cancer Therapy. Cancer Res. 2010;70(9):3718–3729. https://doi.org/10.1158/0008-5....
14.
Jing HX, Duan de J, Zhou H et al. Adipose‑derived mesenchymal stem cell‑facilitated TRAIL expression in melanoma treatment in vitro. Mol Med Rep. 2016;14(1):195–201. https://doi.org/10.3892/mmr.20....
15.
Chen Y, He Y, Wang X, et al. Adipose‑derived mesenchymal stem cells exhibit tumor tropism and promote tumorsphere formation of breast cancer cells. Oncol Rep. 2019;41(4):2126–2136. https://doi.org/10.3892/or.201....
16.
Scioli MG, Storti G, D’Amico F, et al. Adipose-Derived Stem Cells in Cancer Progression: New Perspectives and Opportunities. Int J Mol Sci. 2019;20(13):3296. https://doi.org/10.3390/ijms20....
17.
Cunningham NOM, Ava; and Brougham, Cathy L. The interaction of Adipose Derived Stem Cells and Breast Cancer. SURE Journal: Science Undergraduate Research Experience Journal. 2022;4(1):6.
18.
Kassem DH, Habib SA, Badr OI, et al. Isolation of Rat Adipose Tissue Mesenchymal Stem Cells for Differentiation into Insulin-producing Cells. JoVE. 2022(186):e63348. https://doi.org/10.3791/63348.
19.
Wu Q, He J, Herrler T, et al. Adipose-derived stem cells enhance the tumorigenic potential of pre-malignant breast epithelial cells through paracrine activation of PI3K-AKT pathway. Breast Cancer. 2025;32(3):552–565. https://doi.org/10.1007/s12282....
20.
Ordak M. Implementation of SAMPL guidelines: Recommendations for improving statistical reporting in biomedical journals. Clin Med (Lond). 2025;25(3):100304. https://doi.org/10.1016/j.clin....
21.
Mildmay-White A, Khan W. Cell Surface Markers on Adipose-Derived Stem Cells: A Systematic Review. Curr Stem Cell Res Ther. 2017;12(6):484–492. https://doi.org/10.2174/157488....
22.
Louzada S, Adega F, Chaves R. Defining the sister rat mammary tumor cell lines HH-16 cl.2/1 and HH-16.cl.4 as an in vitro cell model for Erbb2. PLoS One. 2012;7(1):e29923. https://doi.org/10.1371/journa....
23.
Chen F, Zhong X, Dai Q, et al. Human Umbilical Cord MSC Delivered-Soluble TRAIL Inhibits the Proliferation and Promotes Apoptosis of B-ALL Cell In Vitro and In Vivo. Pharmaceuticals. 2022;15(11):1391. https://doi.org/10.3390/ph1511....
24.
Wang Y, Qian X, Wang Y, et al. Turn TRAIL Into Better Anticancer Therapeutic Through TRAIL Fusion Proteins. Cancer Med. 2025;14(1):e70517. https://doi.org/10.1002/cam4.7....
25.
Huang C-C, Cheng Y-C, Lin Y-C, et al. CSC-3436 sensitizes triple negative breast cancer cells to TRAIL-induced apoptosis through ROS-mediated p38/CHOP/death receptor 5 signaling pathways. Environ Toxicol. 2021;36(12):2578–2588. https://doi.org/10.1002/tox.23....
26.
Piechna K, Juszczyński P. Mechanism of activity and defensive strategies of cancer cells against TRAIL-induced apoptosis. Hematol Clin Pract. 2019;10(3):135–147. https://doi.org/10.5603/Hem.20....
27.
Parsons AM, Ahsan N, Darling EM. Identifying Immunomodulatory Subpopulations of Adipose Stromal Vascular Fraction and Stem/Stromal Cells Through Single-Cell Transcriptomics and Bulk Proteomics. Stem Cell Rev Rep. 2025;21(5):1484–1500. https://doi.org/10.1007/s12015....
28.
Mazur S, Zołocińska A, Siennicka K, et al. Safety of adipose-derived cell (stromal vascular fraction – SVF) augmentation for surgical breast reconstruction in cancer patients. Adv Clin Exp Med. 2018; 27 (8): 1085–1090. https://doi.org/10.17219/acem/....
29.
Ritter A, Friemel A, Fornoff F, et al. Characterization of adipose-derived stem cells from subcutaneous and visceral adipose tissues and their function in breast cancer cells. Oncotarget. 2015;6(33):34475–34493. https://doi.org/10.18632/oncot....
30.
Trevor LV, Riches-Suman K, Mahajan AL, et al. Stromal Vascular Fraction Cells from Individuals Who Have Previously Undergone Radiotherapy Retain Their Pro-Wound Healing Properties. Journal of Clinical Medicine. 2023;12(5):2052. https://doi.org/10.3390/jcm120....
Share
RELATED ARTICLE
We process personal data collected when visiting the website. The function of obtaining information about users and their behavior is carried out by voluntarily entered information in forms and saving cookies in end devices. Data, including cookies, are used to provide services, improve the user experience and to analyze the traffic in accordance with the Privacy policy. Data are also collected and processed by Google Analytics tool (more).
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.