Online first
About the Journal
Current issue
Archive
Publication Ethics
Anti-Plagiarism system
Instructions for Authors
Instructions for Reviewers
Editorial Office
Editorial Board
Contact
Reviewers
All Reviewers
2025
2024
2023
2022
2021
2020
2019
2018
2017
2016
General Data Protection Regulation (RODO)
REVIEW PAPER
Negative effect of general anesthesia on the human brain - mechanism and methods of prevention
1
Student Scientific Society, Department and I Clinic of Anaesthesiology and Intensive Therapy, Medical University, Lublin, Poland
2
Department and I Clinic of Anaesthesiology and Intensive Therapy, Medical University, Lublin, Poland
Corresponding author
Aleksandra Dembowska
Student Scientific Society, Department and I Clinic of Anaesthesiology and Intensive Therapy, Medical University, Lublin, Poland
Student Scientific Society, Department and I Clinic of Anaesthesiology and Intensive Therapy, Medical University, Lublin, Poland
J Pre Clin Clin Res. 2023;17(4):245-250
KEYWORDS
TOPICS
ABSTRACT
Introduction and objective:
General anesthesia is an integral part of many medical procedures. Its main premise is that it is completely reversible and has no effect on the human brain. Unfortunately, a number of scientific findings report potential effects of anaesthetics on the human nervous system. The aim of this review is to present the negative impact of sevoflurane (inhalation anaesthetic) and propofol (intravenous anaesthetic) on the human brain with an analysis of its mechanisms and factors, and possible methods of preventing this phenomenon.
Review methods:
A review was undertaken of the most current literature using PubMed and Google Scholar databases, on the mechanism of action and the negative effects of sevoflurane and propofol on the brain and methods of its prevention. Included are both original works, reviews and case reports from Poland and worldwide.
Brief description of the state of knowledge:
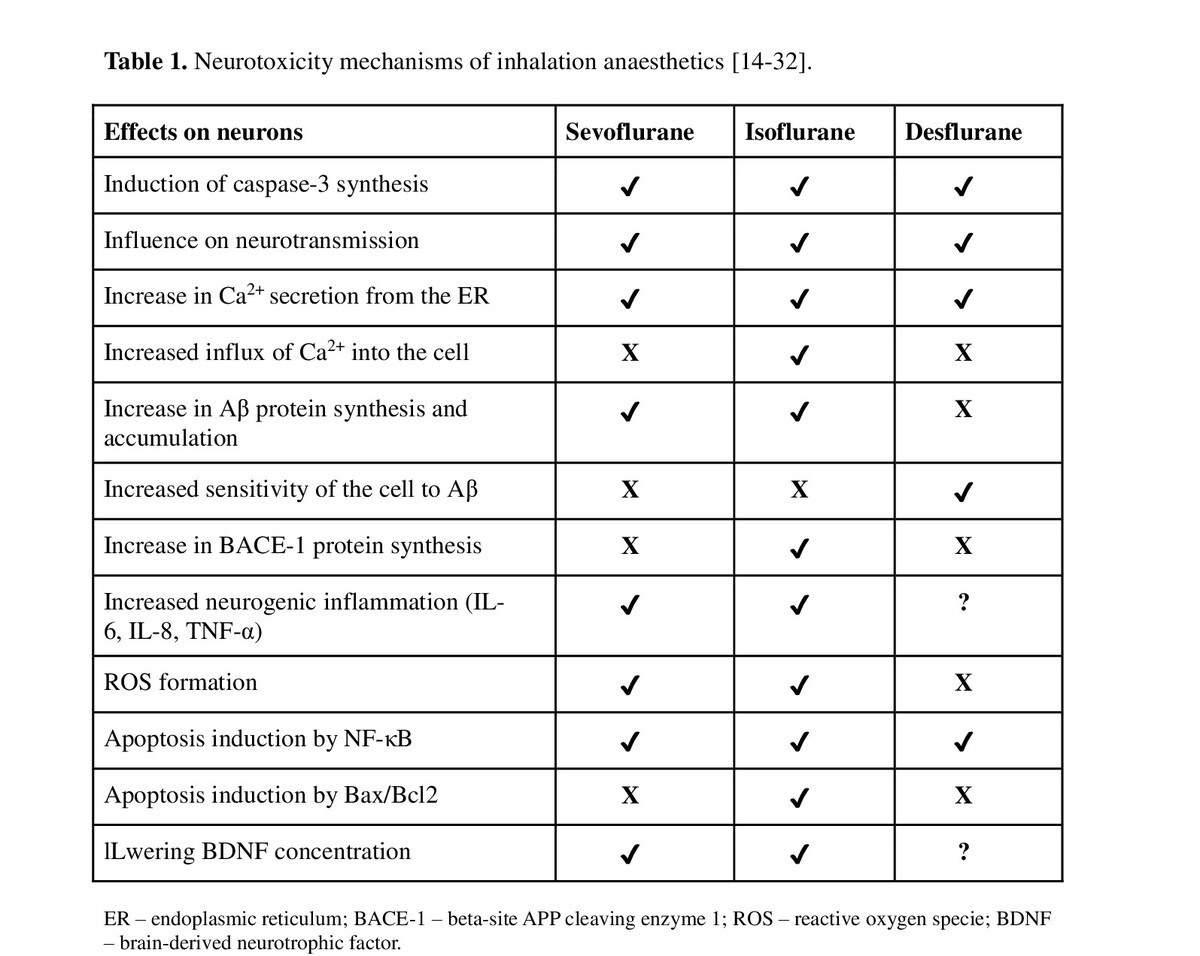
Post-operative cognitive dysfunction affects 10–65% of patients. Sevoflurane and propofol have potentially damaging effects on the human brain. This occurs mainly through induction of neuronal cell apoptosis and disruption of synaprogenesis by affecting receptor proteins, cell organelles (mitochondria, endoplasmic reituclum) and signaling pathways (apoptotic and trophic) of neurons. However, this effect depends on the drug dose and patient-dependent factors (age, diseases, cognitive status before anaesthesia).
Summary:
Anaesthetics have potentially harmful effects on the human brain, although only in large and repeated doses. One should also not forget the much more notable effect of mechanical damage to tissues during surgery. In order to prevent cognitive dysfunction, both a detailed preoperative assessment of the patient’s condition and the use of additional pharmacological and non-pharmacological measures during surgery are important.
General anesthesia is an integral part of many medical procedures. Its main premise is that it is completely reversible and has no effect on the human brain. Unfortunately, a number of scientific findings report potential effects of anaesthetics on the human nervous system. The aim of this review is to present the negative impact of sevoflurane (inhalation anaesthetic) and propofol (intravenous anaesthetic) on the human brain with an analysis of its mechanisms and factors, and possible methods of preventing this phenomenon.
Review methods:
A review was undertaken of the most current literature using PubMed and Google Scholar databases, on the mechanism of action and the negative effects of sevoflurane and propofol on the brain and methods of its prevention. Included are both original works, reviews and case reports from Poland and worldwide.
Brief description of the state of knowledge:
Post-operative cognitive dysfunction affects 10–65% of patients. Sevoflurane and propofol have potentially damaging effects on the human brain. This occurs mainly through induction of neuronal cell apoptosis and disruption of synaprogenesis by affecting receptor proteins, cell organelles (mitochondria, endoplasmic reituclum) and signaling pathways (apoptotic and trophic) of neurons. However, this effect depends on the drug dose and patient-dependent factors (age, diseases, cognitive status before anaesthesia).
Summary:
Anaesthetics have potentially harmful effects on the human brain, although only in large and repeated doses. One should also not forget the much more notable effect of mechanical damage to tissues during surgery. In order to prevent cognitive dysfunction, both a detailed preoperative assessment of the patient’s condition and the use of additional pharmacological and non-pharmacological measures during surgery are important.
Dembowska A, Dubaj M, Bigosiński K, Rutyna R. Negative effect of general anaesthesia on the human brain – mechanism and methods of
prevention. J Pre-Clin Clin Res. 2023; 17(4): 245–250. doi: 10.26444/jpccr/172967
REFERENCES (48)
1.
Lenhardt R. Body temperature regulation and anaesthesia. Handb Clin Neurol. 2018;157:635–644. doi: 10.1016/B978–0–444–64074–1.00037–9.
2.
Abdoli J, Motamedi SA, Zargaran A. A Short Review on the History of Anaesthesia in Ancient Civilizations. Journal of Research on History of Medicine, 2019;8(3): 147–156.
3.
Bartkowska-Śniatkowska A, Rosada-Kurasińska J, Zielińska M, Bienert A. Do we really know pharmacodynamics of anaesthetics used in newborns, infants and children? A review of experimental and clinical data on neurodegeneration. Anestezjologia Intensywna Terapia. 2014:111–118. ISSN 0209–1712.
4.
Armstrong R, Xu F, Arora A, et al. General anesthetics and cytotoxicity: possible implications for brain health. Drug Chem Toxicol. 2017 Apr;40(2):241–249. doi: 10.1080/01480545.2016.1188306.
5.
Palanca BJA, Avidan MS, Mashour GA. Human neural correlates of sevoflurane-induced unconsciousness. Br J Anaesth. 2017;119(4):573–582. doi: 10.1093/bja/aex244.
6.
Brohan J, Goudra BG. The Role of GABA Receptor Agonists in Anaesthesia and Sedation. CNS Drugs. 2017;31(10):845–856. doi: 10.1007/s40263–017–0463–7.
7.
Walsh CT. Propofol: Milk of Amnesia. Cell. 2018;175(1):10–13. doi: 10.1016/j.cell.2018.08.031.
8.
Vyklicky V, Korinek M, Smejkalova T, et al. Structure, function, and pharmacology of NMDA receptor channels. Physiol Res. 2014;63(Suppl 1):S191–203. doi: 10.33549/physiolres.932678.
9.
Changeux JP. The nicotinic acetylcholine receptor: the founding father of the pentameric ligand-gated ion channel superfamily. J Biol Chem. 2012;287(48):40207–15. doi: 10.1074/jbc.R112.407668.
10.
Changeux JP. Discovery of the First Neurotransmitter Receptor: The Acetylcholine Nicotinic Receptor. Biomolecules. 2020;10(4):547. doi: 10.3390/biom10040547.
11.
Huang JM, Lv ZT, Zhang B, et al. Intravenous parecoxib for early postoperative cognitive dysfunction in elderly patients: evidence from a meta-analysis. Expert Rev Clin Pharmacol. 2020;13(4):451–460. doi: 10.1080/17512433.2020.1732815.
12.
Boone MD, Sites B, von Recklinghausen FM, et al. Economic Burden of Postoperative Neurocognitive Disorders Among US Medicare Patients. JAMA Netw Open. 2020;3(7):e208931. doi: 10.1001/jamanetworkopen.2020.8931.
13.
Evered L, Silbert B, Knopman DS, et al. Recommendations for the Nomenclature of Cognitive Change Associated with Anaesthesia and Surgery-2018. Anesthesiology. 2018 Nov;129(5):872–879. doi: 10.1097/ALN.0000000000002334.
14.
Wang CM, Chen WC, Zhang Y, et al. Update on the Mechanism and Treatment of Sevoflurane-Induced Postoperative Cognitive Dysfunction. Front Aging Neurosci. 2021;13:702231. doi: 10.3389/fnagi.2021.702231.
15.
Hovens IB, van Leeuwen BL, Mariani MA, et al. Postoperative cognitive dysfunction and neuroinflammation; Cardiac surgery and abdominal surgery are not the same. Brain Behav Immun. 2016;54:178–193. doi: 10.1016/j.bbi.2016.02.003.
16.
Cui RS, Wang K, Wang ZL. Sevoflurane anaesthesia alters cognitive function by activating inflammation and cell death in rats. Exp Ther Med. 2018;15(5):4127–4130. doi: 10.3892/etm.2018.5976.
17.
Wei H, Xie Z. Anaesthesia, calcium homeostasis and Alzheimer’s disease. Curr Alzheimer Res. 2009;6(1):30–5. doi: 10.2174/156720509787313934.
18.
Zhang L, Zhang J, Yang L, et al. Isoflurane and sevoflurane increase interleukin-6 levels through the nuclear factor-kappa B pathway in neuroglioma cells. Br J Anaesth. 2013;110 Suppl 1(Suppl 1):i82–91. doi: 10.1093/bja/aet115.
19.
Zhu X, Yao Y, Guo M, et al. Sevoflurane increases intracellular calcium to induce mitochondrial injury and neuroapoptosis. Toxicol Lett. 2021;336:11–20. doi: 10.1016/j.toxlet.2020.11.002.
20.
Xu Z, Qian B. Sevoflurane anaesthesia-mediated oxidative stress and cognitive impairment in hippocampal neurons of old rats can be ameliorated by expression of brain derived neurotrophic factor. Neurosci Lett. 2020;721:134785. doi: 10.1016/j.neulet.2020.134785.
21.
Yang N, Li Z, Han D, et al. Autophagy prevents hippocampal α-synuclein oligomerization and early cognitive dysfunction after anaesthesia/surgery in aged rats. Aging (Albany NY). 2020;12(8):7262–7281. doi: 10.18632/aging.103074.
22.
Apai C, Shah R, Tran K, Pandya Shah S. Anaesthesia and the Developing Brain: A Review of Sevoflurane-induced Neurotoxicity in Pediatric Populations. Clin Ther. 2021;43(4):762–778. doi: 10.1016/j.clinthera.2021.01.024.
23.
Neag MA, Mitre AO, Catinean A, Mitre CI. An Overview on the Mechanisms of Neuroprotection and Neurotoxicity of Isoflurane and Sevoflurane in Experimental Studies. Brain Res Bull. 2020;165:281–289. doi: 10.1016/j.brainresbull.2020.10.011.
24.
Meineke M, Applegate RL 2nd, Rasmussen T, et al. Cognitive dysfunction following desflurane versus sevoflurane general anaesthesia in elderly patients: a randomized controlled trial. Med Gas Res. 2014;4(1):6. doi: 10.1186/2045–9912–4–6.
25.
Liu XX, Yang QX, Guo Y, et al. Cognitive impairment induced by sevoflurane anaesthesia is mediated by the cholinergic system after gastrointestinal surgery in older patients: A randomized, controlled trial. Ibrain. 2022:1–9. doi: 10.1002/ibra.12079.
26.
Zhang B, Tian M, Zhen Y, et al. The effects of isoflurane and desflurane on cognitive function in humans. Anesth Analg. 2012;114(2):410–5. doi: 10.1213/ANE.0b013e31823b2602.
27.
Jiang J, Jiang H. Effect of the inhaled anesthetics isoflurane, sevoflurane and desflurane on the neuropathogenesis of Alzheimer’s disease (review). Mol Med Rep. 2015;12(1):3–12. doi: 10.3892/mmr.2015.3424.
28.
Zhang S, Hu X, Guan W, et al. Isoflurane anaesthesia promotes cognitive impairment by inducing expression of β-amyloid protein-related factors in the hippocampus of aged rats. PLoS One. 2017;12(4):e0175654. doi: 10.1371/journal.pone.0175654.
29.
Xiao XL, Wu JT, Zhang HZ, et al. The neurotoxic effect of isoflurane on age-defined neurons generated from tertiary dentate matrix in mice. Brain Behav. 2021;11(1):e01949. doi: 10.1002/brb3.1949.
30.
Zhang Y, Dong Y, Wu X, et al. The mitochondrial pathway of anesthetic isoflurane-induced apoptosis. J Biol Chem. 2010;285(6):4025–4037. doi: 10.1074/jbc.M109.065664.
31.
Rörtgen D, Kloos J, Fries M, et al. Comparison of early cognitive function and recovery after desflurane or sevoflurane anaesthesia in the elderly: a double-blinded randomized controlled trial. Br J Anaesth.2010;104(2):167–74. doi: 10.1093/bja/aep369.
32.
Niikura R, Miyazaki T, Takase K, et al. Assessments of prolonged effects of desflurane and sevoflurane on motor learning deficits in aged AppNL-G-F/NL-G-F mice. Mol Brain. 2022;15(1):32. doi: 10.1186/ s13041–022–00910–1.
33.
Berndt N, Rösner J, Haq RU, et al. Possible neurotoxicity of the anesthetic propofol: evidence for the inhibition of complex II of the respiratory chain in area CA3 of rat hippocampal slices. Arch Toxicol. 2018 Oct;92(10):3191–3205. doi: 10.1007/s00204–018–2295–8.
34.
Hou JF, Xiao CL. Effect of propofol and sevoflurane anaesthesia on postoperative cognitive function and levels of Aβ-42 and Tau in patients undergoing hepatectomy. Eur Rev Med Pharmacol Sci. 2019;23(2):849– 856. doi: 10.26355/eurrev_201901_16900.
35.
Goswami U, Babbar S, Tiwari S. Comparative evaluation of the effects of propofol and sevoflurane on cognitive function and memory in patients undergoing laparoscopic cholecystectomy: A randomised prospective study. Indian J Anaesth. 2015;59(3):150–5. doi: 10.4103/0019– 5049.153036.
36.
Konishi Y, Evered LA, Scott DA, Silbert BS. Postoperative cognitive dysfunction after sevoflurane or propofol general anaesthesia in combination with spinal anaesthesia for hip arthroplasty. Anaesth Intensive Care. 2018;46(6):596–600. doi: 10.1177/0310057X1804600610.
37.
Kim JH, Jung H, Lee Y, Sohn JH. Surgery Performed Under Propofol Anaesthesia Induces Cognitive Impairment and Amyloid Pathology in ApoE4 Knock-In Mouse Model. Front Aging Neurosci. 2021;13:658860. doi: 10.3389/fnagi.2021.658860.
38.
Han D, Jin J, Fang H, Xu G. Long-term action of propofol on cognitive function and hippocampal neuroapoptosis in neonatal rats. Int J Clin Exp Med. 2015;8(7):10696–704.
39.
Berger M, Schenning KJ, Brown CH 4th, et al. Perioperative Neurotoxicity Working Group. Best Practices for Postoperative Brain. Health: Recommendations From the Fifth International Perioperative Neurotoxicity Working Group. Anesth Analg. 2018;127(6):1406–1413. doi: 10.1213/ANE.0000000000003841.
40.
Culley DJ, Flaherty D, Reddy S, et al. Preoperative Cognitive Stratification of Older Elective Surgical Patients: A Cross-Sectional Study. Anesth Analg. 2016;123(1):186–92. doi: 10.1213/ANE.0000000000001277.
41.
Mapleson WW. Effect of age on MAC in humans: a meta-analysis. Br J Anaesth. 1996;76(2):179–85. doi: 10.1093/bja/76.2.179.
42.
Chan MT, Cheng BC, Lee TM, Gin T; CODA Trial Group. BISguided anaesthesia decreases postoperative delirium and cognitive decline. J Neurosurg Anesthesiol. 2013;25(1):33–42. doi: 10.1097/ ANA.0b013e3182712fba.
43.
Ikonomidou C, Kirvassilis G, Swiney BS, et al. Mild hypothermia ameliorates anaesthesia toxicity in the neonatal macaque brain. Neurobiol Dis. 2019;130:104489. doi: 10.1016/j.nbd.2019.104489.
44.
Yocum GT, Gaudet JG, Teverbaugh LA, et al. Neurocognitive performance in hypertensive patients after spine surgery. Anesthesiology. 2009;110(2):254–61. doi: 10.1097/ALN.0b013e3181942c7a.
45.
Zheng F, Sheinberg R, Yee MS, et al. Cerebral near-infrared spectroscopy monitoring and neurologic outcomes in adult cardiac surgery patients: a systematic review. Anesth Analg. 2013;116(3):663–76. doi: 10.1213/ ANE.0b013e318277a255.
46.
Kim J, Shim JK, Song JW, et al. Postoperative Cognitive Dysfunction and the Change of Regional Cerebral Oxygen Saturation in Elderly Patients Undergoing Spinal Surgery. Anesth Analg. 2016;123(2):436–44. doi: 10.1213/ANE.0000000000001352.
47.
Viderman D, Aubakirova M, Nabidollayeva F, et al. Effect of Ketamine on Postoperative Neurocognitive Disorders: A Systematic Review and Meta-Analysis. J Clin Med. 2023;12(13):4314. Published 2023 Jun 27. doi:10.3390/jcm12134314.
48.
Lemoine S, Zhu L, Gérard JL, Hanouz JL. Sevoflurane-induced cardioprotection in coronary artery bypass graft surgery: Randomised trial with clinical and ex-vivo endpoints. Anaesth Crit Care Pain Med. 2018;37(3):217–223. doi:10.1016/j.ac.
Share
RELATED ARTICLE
We process personal data collected when visiting the website. The function of obtaining information about users and their behavior is carried out by voluntarily entered information in forms and saving cookies in end devices. Data, including cookies, are used to provide services, improve the user experience and to analyze the traffic in accordance with the Privacy policy. Data are also collected and processed by Google Analytics tool (more).
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.