Online first
About the Journal
Current issue
Archive
Publication Ethics
Anti-Plagiarism system
Instructions for Authors
Instructions for Reviewers
Editorial Office
Editorial Board
Contact
Reviewers
All Reviewers
2025
2024
2023
2022
2021
2020
2019
2018
2017
2016
General Data Protection Regulation (RODO)
RESEARCH PAPER
Interactions of C-11 with selected antiseizure medications in the mouse 6 Hz psychomotor seizure model – an isobolographic analysis
1
Department of Experimental Pharmacology, Institute of Rural Health, Lublin, Poland
2
Department of Occupational Medicine, Medical University, Lublin, Poland
3
Department of Medicinal Chemistry, Jagiellonian University Medical College, Kraków, Poland
Corresponding author
J Pre Clin Clin Res. 2026;20(1):1-4
KEYWORDS
TOPICS
ABSTRACT
Introduction and objective:
Currently, polytherapy with two or more antiseizure medications (ASMs) remains one of the therapeutic options for patients with drug-resistant epilepsy. C-11, a pyrrolidine-2,5-dione derivative with previously demonstrated antiseizure activity, has been proposed as a potential adjunct compound. The aim of the present study is to characterize the type of interactions between C-11 and two clinically used ASMs, lacosamide (LCM) and valproate (VPA), in the mouse psychomotor (6 Hz, 32 mA) seizure model.
Material and methods:
In the 6 Hz seizure model, anticonvulsant activity was evaluated in male albino Swiss mice by assessing protection against psychomotor seizures. Dose–response relationships were established using log–probit analysis, and median effective doses (ED50 ± SEM) were calculated. Type I isobolographic analysis was applied to determine the interactions between C-11 and LCM or VPA administered in a fixed-ratio combination (1:1).
Results:
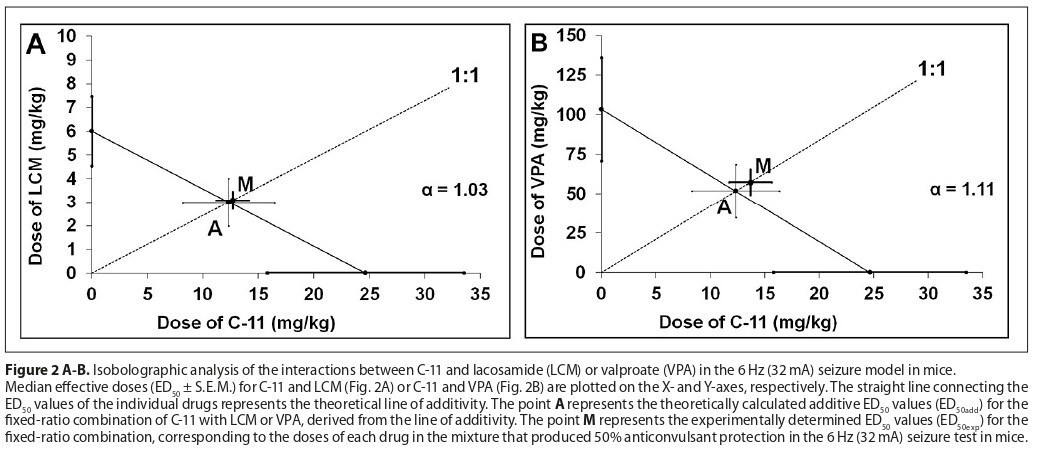
Log–probit analysis demonstrated that C-11, LCM, and VPA administered separately produced clear anticonvulsant effects in the 6 Hz seizure model in mice, and linear regression confirmed parallel dose–response relationships for C-11 in combination with selected ASMs. Type I isobolographic analysis demonstrated that C-11 combined with LCM or VPA (fixedratio of 1:1) produced additive interactions in the 6 Hz model.
Conclusions:
The study shows that mixtures of C-11 with LCM or VPA, evaluated by isobolographic analysis, produce additive interactions in the 6 Hz (32 mA) seizure test in mice. Further studies are required to clarify the mechanisms underlying these effects and to determine their potential clinical relevance.
Currently, polytherapy with two or more antiseizure medications (ASMs) remains one of the therapeutic options for patients with drug-resistant epilepsy. C-11, a pyrrolidine-2,5-dione derivative with previously demonstrated antiseizure activity, has been proposed as a potential adjunct compound. The aim of the present study is to characterize the type of interactions between C-11 and two clinically used ASMs, lacosamide (LCM) and valproate (VPA), in the mouse psychomotor (6 Hz, 32 mA) seizure model.
Material and methods:
In the 6 Hz seizure model, anticonvulsant activity was evaluated in male albino Swiss mice by assessing protection against psychomotor seizures. Dose–response relationships were established using log–probit analysis, and median effective doses (ED50 ± SEM) were calculated. Type I isobolographic analysis was applied to determine the interactions between C-11 and LCM or VPA administered in a fixed-ratio combination (1:1).
Results:
Log–probit analysis demonstrated that C-11, LCM, and VPA administered separately produced clear anticonvulsant effects in the 6 Hz seizure model in mice, and linear regression confirmed parallel dose–response relationships for C-11 in combination with selected ASMs. Type I isobolographic analysis demonstrated that C-11 combined with LCM or VPA (fixedratio of 1:1) produced additive interactions in the 6 Hz model.
Conclusions:
The study shows that mixtures of C-11 with LCM or VPA, evaluated by isobolographic analysis, produce additive interactions in the 6 Hz (32 mA) seizure test in mice. Further studies are required to clarify the mechanisms underlying these effects and to determine their potential clinical relevance.
Zagaja M, Szewczyk A, Szala-Rycaj J, Łuszczki J, Kamiński K, Andres-Mach M. Interactions of C-11 with selected antiseizure medications in
the mouse 6 Hz psychomotor seizure model – an isobolographic analysis. J Pre-Clin Clin Res. 2026;20(1):1–4. doi:10.26444/jpccr/219040
REFERENCES (26)
1.
Beghi E. The Epidemiology of Epilepsy. Neuroepidemiology. 2020;54(2):185–191.
2.
Löscher W, Potschka H, Sisodiya SM, Vezzani A. Drug Resistance in Epilepsy: Clinical Impact, Potential Mechanisms, and New Innovative Treatment Options. Pharmacol Rev. 2020;72(3):606–638.
3.
Rai D, Kerr MP, McManus S, Jordanova V, Lewis G, Brugha TS. Epilepsy and psychiatric comorbidity: a nationally representative population-based study. Epilepsia, 2012;53:1095–1103.
4.
Stephen LJ, Brodie MJ. Antiepileptic drug monotherapy versus polytherapy: Pursuing seizure freedom and tolerability in adults. Curr. Opin. Neurol. 2012;25:164–172.
5.
Zagaja M, Pyrka D, Skalicka-Wozniak K, Glowniak K, Florek-Luszczki M, Glensk M, Luszczki JJ. Effect of xanthotoxin (8-methoxypsoralen) on the anticonvulsant activity of classical antiepileptic drugs against maximal electroshock-induced seizures in mice. Fitoterapia. 2015;105:1–6.
6.
Zagaja M, Andres-Mach M, Skalicka-Woźniak K, Rękas AR, Kondrat-Wróbel MW, Gleńsk M, Łuszczki JJ. Assessment of the Combined Treatment with Umbelliferone and Four Classical Antiepileptic Drugs Against Maximal Electroshock-Induced Seizures in Mice. Pharmacology. 2015;96(3–4):175–180.
7.
Zagaja M, Andres-Mach M, Patrzylas P, Pyrka D, Szpringer M, Florek-Łuszczki M, Żółkowska D, Skalicka-Woźniak K, Łuszczki JJ. Influence of xanthotoxin (8-methoxypsoralen) on the anticonvulsant activity of various novel antiepileptic drugs against maximal electroshock-induced seizures in mice. Fitoterapia. 2016;115:86–91.
8.
Kamiński K, Zagaja M, Łuszczki JJ, Rapacz A, Andres-Mach M, Latacz G, Kieć- Kononowicz K. Design, synthesis, and anticonvulsant activity of new hybrid compounds derived from 2-(2,5-dioxopyrrolidin-1-yl) propanamides and 2-(2,5 dioxopyrrolidin-1-yl) butanamides. J. Med. Chem. 2015;58:5274–5286.
9.
Zagaja M, Szewczyk A, Szala-Rycaj J, Raszewski G, Chrościńska-Krawczyk M, Abram M, Kamiński K, Andres-Mach M. C-11, a New Antiepileptic Drug Candidate: Evaluation of the Physicochemical Properties and Impact on the Protective Action of Selected Antiepileptic Drugs in the Mouse Maximal Electroshock-Induced Seizure Model. Molecules. 2021;26(11):3144..
10.
Socała K, Mogilski S, Pieróg M, Nieoczym D, Abram M, Szulczyk B, Lubelska A, Latacz G, Doboszewska U, Wlaź P, Kamiński K. KA-11, a Novel Pyrrolidine-2,5-dione Derived Broad-Spectrum Anticonvulsant: Its Antiepileptogenic, Antinociceptive Properties and in Vitro Characterization. ACS Chem Neurosci. 2019;10(1):636–648.
11.
Barton ME, Klein BD, Wolf HH, White HS. Pharmacological characterization of the 6 Hz psychomotor seizure model of partial epilepsy. Epilepsy Res. 2001;47:217–227.
12.
Barton ME, Peters SC, Shannon HE. Comparison of the effect of glutamate receptor modulators in the 6 Hz and maximal electroshock seizure models. Epilepsy Res. 2003;56:17—26.
13.
Zagaja M, Bryda J, Szewczyk A, Szala-Rycaj J, Łuszczki JJ, Walczak M, Kuś K, Andres-Mach M. Xanthotoxin enhances the anticonvulsant potency of levetiracetam and valproate in the 6-Hz corneal stimulation model in mice. Fundam Clin Pharmacol. 2022;36(1):133–142.
14.
Zagaja M, Zagaja A, Szala-Rycaj J, Szewczyk A, Lemieszek MK, Raszewski G, Andres-Mach M. Influence of Umbelliferone on the Anticonvulsant and Neuroprotective Activity of Selected Antiepileptic Drugs: An In Vivo and In Vitro Study. Int J Mol Sci. 2022;23(7):3492.
15.
Żółkowska D, Zagaja M, Miziak B, Kondrat-Wróbel MW, Załuska K, Florek-Łuszczki M, et al. Isobolographic assessment of interactions between retigabine and phenytoin in the mouse maximal electroshock-induced seizure model and chimney test. Health Prob Civil. 2016;10: 54–59.
16.
Zagaja M, Miziak B, Kondrat-Wróbel MW, Andres-Mach M, Wróblewska-Łuczka P, Adamczuk P, Chmura R, Czuczwar SJ, Łuszczki JJ. Interactions of retigabine with topiramate in the mouse tonic-clonic seizure model and chimney test – an isobolographic analysis. Journal of Pre-Clinical and Clinical Research. 2017;11:61–65.
17.
Luszczki JJ, Czuczwar SJ. Preclinical profile of combinations of some second-generation antiepileptic drugs: an isobolographic analysis. Epilepsia. 2004;45:895–907.
18.
Luszczki JJ. Isobolographic analysis of interaction between drugs with nonparallel dose-response relationship curves: a practical application. Naunyn Schmiedebergs Arch Pharmacol. 2007;375:105–114.
19.
Litchfield JT, Jr., Wilcoxon F. A simplified method of evaluating dose-effect experiments. J Pharmacol Exp Ther. 1949;96:99–113.
20.
Tallarida RJ. Revisiting the isobole and related quantitative methods for assessing drug synergism. J Pharmacol Exp Ther. 2012;342:2–8.
21.
Rogawski MA, Tofighy A, White HS, Matagne A, Wolff C. Current understanding of the mechanism of action of the antiepileptic drug lacosamide. Epilepsy Res. 2015;110:189–205.
22.
Wilson SM, Khanna R. Specific binding of lacosamide to collapsin response mediator protein 2 (CRMP2) and direct impairment of its canonical function: Implications for the therapeutic potential of lacosamide. Mol. Neurobiol. 2015;51:599–609.
23.
Löscher W. Basic pharmacology of valproate: a review after 35 years of clinical use for the treatment of epilepsy. CNS Drugs. 2002;16(10):669–694.
24.
Englund M, Hyllienmark L, Brismar T. Effect of valproate, lamotrigine and levetiracetam on excitability and firing properties of CA1 neurons in rat brain slices. Cell Mol Neurobiol. 2011;31(4):645–652.
25.
Metcalf CS, West PJ, Thomson KE, Edwards SF, Smith MD, White HS, Wilcox KS. Development and pharmacologic characterization of the rat 6 Hz model of partial seizures. Epilepsia. 2017;58(6):1073–1084.
26.
Löscher W, White HS. Animal Models of Drug-Resistant Epilepsy as Tools for Deciphering the Cellular and Molecular Mechanisms of Pharmacoresistance and Discovering More Effective Treatments. Cells. 2023;12(9):1233.
Share
RELATED ARTICLE
We process personal data collected when visiting the website. The function of obtaining information about users and their behavior is carried out by voluntarily entered information in forms and saving cookies in end devices. Data, including cookies, are used to provide services, improve the user experience and to analyze the traffic in accordance with the Privacy policy. Data are also collected and processed by Google Analytics tool (more).
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.