Online first
About the Journal
Current issue
Archive
Publication Ethics
Anti-Plagiarism system
Instructions for Authors
Instructions for Reviewers
Editorial Office
Editorial Board
Contact
Reviewers
All Reviewers
2025
2024
2023
2022
2021
2020
2019
2018
2017
2016
General Data Protection Regulation (RODO)
RESEARCH PAPER
Buccal epithelial cells as non-invasive biological material for fibrodysplasia ossificans progressiva gene expression studies
1
Universidade Federal de Mato Grosso do Sul, Brazil
2
Instituto de Assistência em Pesquisa, Educação e Saúde – Iapes, Brazil
3
Universidade Federal de Minas Gerais, Brazil
4
Procter and Gamble Health, USA
Corresponding author
J Pre Clin Clin Res. 2024;18(2):89-94
KEYWORDS
buccal mucosaepithelial cellsFibrodysplasia Ossificans ProgressivaFOP mRNA expressionsquamous cell scrapings
TOPICS
ABSTRACT
Introduction and objective:
The congenital disease Fibrodysplasia Ossificans Progressiva (FOP) is extremely rare, characterized by irreversible and intractable skeletal malformations, with devastating heterotopic ossifications. FOP is associated with a change in the amino acid of the ACVR1 protein at position R206H due to a mutation in its respective genetic code. Previous research has observed distinct gene expression profiles between FOP peripheral blood mononuclear cells (PBMC) versus control cells. However, invasive access to biological material in these FOP patients is an obstacle due to possible collection trauma that can cause flare-ups, with undesirable consequences, such as the formation of ectopic ossification. There is, however, a need to obtain biological specimens for research or monitoring of experimental medications. The aim of the study was to obtain total RNA from buccal mucosa epithelial squamous cell scrapings (BEC) from FOP patients.
Material and methods:
BEC samples were collected by scraping the oral mucosa on the inner side of each cheek, using tongue depressor spatulas, followed by stabilization in RNAlater and Trizol extraction of total RNA. The expression profile of eight putative target Genes were analyzed from FOP BEC (n=7) and healthy volunteers (n=5), by mRNA expression through qPCR.
Results:
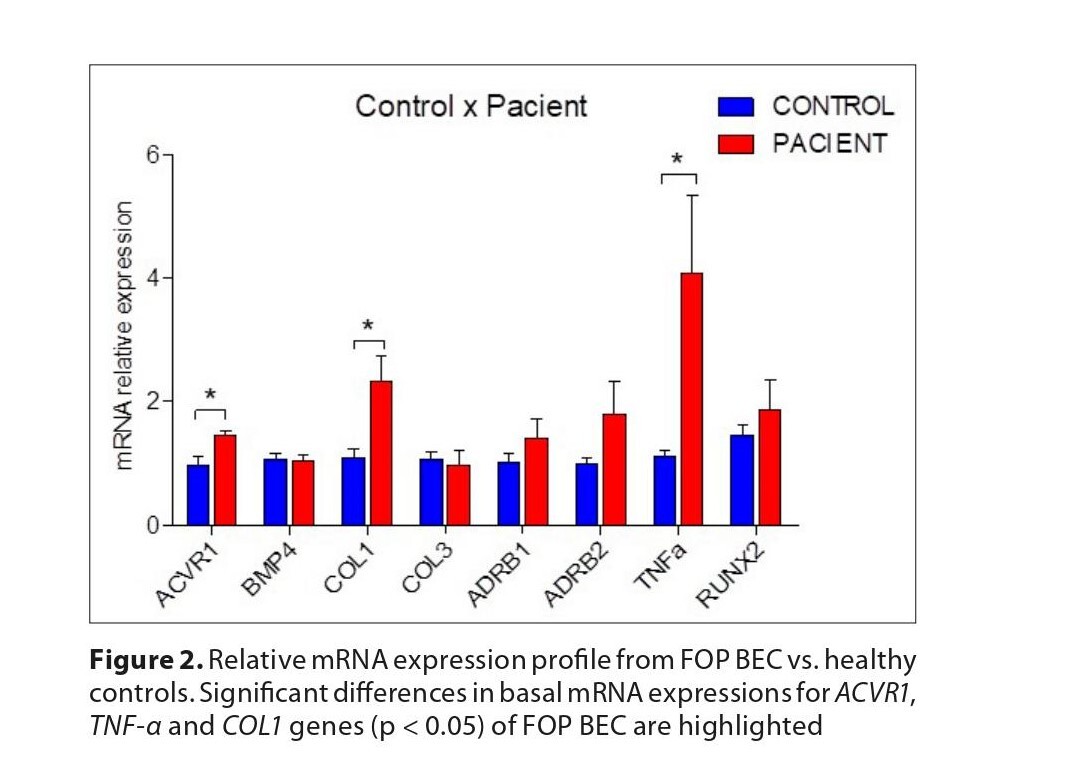
The results showed differences in basal mRNA expression of ACVR1, TNF-α and COL1 (p < 0.05) genes in FOP, compared to control, characterizing a distinct phenotypic profile of FOP BEC.
Conclusions:
The use of BEC may be an innovative non-invasive biological material for further clinical and molecular analyzes in patients with FOP.
The congenital disease Fibrodysplasia Ossificans Progressiva (FOP) is extremely rare, characterized by irreversible and intractable skeletal malformations, with devastating heterotopic ossifications. FOP is associated with a change in the amino acid of the ACVR1 protein at position R206H due to a mutation in its respective genetic code. Previous research has observed distinct gene expression profiles between FOP peripheral blood mononuclear cells (PBMC) versus control cells. However, invasive access to biological material in these FOP patients is an obstacle due to possible collection trauma that can cause flare-ups, with undesirable consequences, such as the formation of ectopic ossification. There is, however, a need to obtain biological specimens for research or monitoring of experimental medications. The aim of the study was to obtain total RNA from buccal mucosa epithelial squamous cell scrapings (BEC) from FOP patients.
Material and methods:
BEC samples were collected by scraping the oral mucosa on the inner side of each cheek, using tongue depressor spatulas, followed by stabilization in RNAlater and Trizol extraction of total RNA. The expression profile of eight putative target Genes were analyzed from FOP BEC (n=7) and healthy volunteers (n=5), by mRNA expression through qPCR.
Results:
The results showed differences in basal mRNA expression of ACVR1, TNF-α and COL1 (p < 0.05) genes in FOP, compared to control, characterizing a distinct phenotypic profile of FOP BEC.
Conclusions:
The use of BEC may be an innovative non-invasive biological material for further clinical and molecular analyzes in patients with FOP.
Ana Ligia Barbosa Messias, Suzana Lopes Bomfim Balaniuc, Lorena Falcão Lima, Deborah Ribeiro Nascimento, Marilene Garcia Palhares,
Fabiana Alves, Helen Lima Del Puerto, Jose Mauro Goulart Brum, Durval Batista Palhares, Almir Sousa Martins. Buccal epithelial cells as
non-invasive biological material for fibrodysplasia ossificans progressiva gene expression studies. J Pre-Clin Clin Res. 2024; 18(2): 89–94.
doi: 10.26444/jpccr/188214
REFERENCES (40)
1.
Pignolo RJ, Baujat G, Brown MA, et al. Natural history of fibrodysplasia ossificans progressiva: cross-sectional analysis of annotated baseline phenotypes. Orphanet J Rare Dis. 2019;14(98):1–11. https://doi:10.1186/s13023-019....
2.
Kaplan FS, Mukaddam MA, Stanley A, et al. Fibrodysplasia ossificans progressiva (FOP): A disorder of osteochondrogenesis. Bone. 2020;140(115539):1–6. https://doi:10.1016/j.bone.202....
3.
Zorzi AR, Delai PR, Rosa HLC, et al. Fibrodysplasia ossificans progressiva in Brazil: challenges and strategies to create assistance and educational networks. Orphanet J Rare Dis. 2022;17(348)1–4. https://doi:10.1186/s13023-022....
4.
Srinivasan D, Arostegui M, Goebel EJ, et al. How Activin A Became a Therapeutic Target in Fibrodysplasia Ossificans Progressiva. Biomolecules. 2024;14(101):1–14 https://doi:10.3390/biom140101....
5.
Barruet E, Garcia SM, Wu J, et al. Modeling the ACVR1R206H mutation in human skeletal muscle stem cells. eLife. 2021;0(e66107):1–25. https://doi.org/10.7554/eLife.....
6.
Valer JA, Sánchez-de-Diego C, Pimenta-Lopes C, et al. ACVR1 function in health and disease. Cells. 2019;8(1366):1–26. https://doi.org/10.3390/cells8....
7.
Ravazzolo R, Bocciardi R. Genomic Context and Mechanisms of the ACVR1 Mutation in Fibrodysplasia Ossificans Progressiva. Biomedicines. 2021;9(154):1–12. https://doi.org/10.3390/biomed....
8.
Anwar S, Yokota T. Navigating the Complex Landscape of Fibrodysplasia Ossificans Progressiva: From Current Paradigms to Therapeutic Frontiers. Genes. 2023;14(2162):1–25. https://doi.org/10.3390/genes1....
9.
De Brasi D, Orlando F, Gaeta V, et al. Fibrodysplasia Ossificans Progressiva: A Challenging Diagnosis. Genes. 2021;12(1187):1–12. https://doi.org/10.3390/genes1....
10.
Pignolo RJ, Hsiao EC, Baujat G, et al. Prevalence of fbrodysplasia ossifcans progressiva (FOP) in the United States: estimate from three treatment centers and a patient organization. Orphanet J Rare Dis. 2021;16(350):1–8. https://doi.org/10.1186/s13023....
11.
Sun D, Peng Liu P, Wang Z, et al. Fibrodysplasia ossificans progressiva: A rare disease with spinal deformity and severe hip dysfunction. Frontiers in Pediatrics. 2022;10:01–08. https://doi.org/10.3389/fped.2....
12.
Pignolo RJ, Wang H, Kaplan FS. Fibrodysplasia Ossificans Progressiva (FOP): A Segmental Progeroid Syndrome. Frontiers in Endocrinology. 2020;10(908):1–8. https://doi.org/10.3389/fendo.....
13.
Kaplan FS, Zeitlin L, Dunn SP, et al. Acute and chronic rapamycin use in patients with fibrodysplasia ossificans progressiva: a report of two cases. Bone. 2018;109:281–284. https://doi.org/10.1016/j.bone....
14.
Shah ZA, Rausch S, Arif U, et al. Fibrodysplasia ossificans progressiva (stone man syndrome): a case report. Journal of Medical Case Reports. 2019;13(364):1–5. https://doi.org/10.1186/s13256....
15.
Katagiri T, Tsukamoto S, Kuratani M. Heterotopic bone induction via BMP signaling: potencial therapeutic targets for fibrodysplasia ossificans progressiva. Bone. 2018;109:241–250. https://doi.org/10.1016/j.bone....
16.
Wentworth KL, Masharani U, Hsiao EC. Therapeutic advances for blocking heterotopic ossification in fibrodysplasia ossificans progressiva. Br J Clin Pharmacol. 2019;85(6):1180–1187. https://doi.org/10.1111/bcp.13...
17.
Lees-Shepard JB, E Nicholas SA, Stoessel SJ, et al. Palovarotene reduces heterotopic ossification in juvenile FOP mice but exhibits pronounced skeletal toxicity. eLife. 2018;7(e40814):1–20. https://doi.org/10.7554/eLife.....
18.
Palhares DB, Nascimento DR, Palhares MG, et al. Propranolol and ascorbic acid in control of fibrodysplasia ossificans progressiva flare-ups due to accidental falls. Intractable and Rare Diseases Research. 2019;8(1):24–28. https://doi.org/10.5582/irdr.2....
19.
Balaniuc SLB, Nascimento DR, Palhares DB, et al. Fibrodysplasia Ossificans Progressiva: pro-inflammatory and profibrotic genes as targets for research and treatment of clinical symptoms of this rare and 55 devastating disease. Revista NBC. 2019;9(17):1–14.
20.
Nascimento DR, Palhares DB, Del Puerto HL, et al. Ascorbic acid modulates the expression of genes involved in heterotopic ossification. NBC-Periódico Científico do Núcleo de Biociências. 2017;7(14):81–97.
21.
Nascimento DR, Balaniuc SLB, Palhares DB, et al. Rare and intractable fibrodysplasia ossificans progressiva shows different PBMC phenotype possibly modulated by ascorbic acid and propranolol treatment. Intractable Rare Dis Res. 2021;10(3):179–189. https://doi.org/10.5582/irdr.2....
22.
Barruet E, Morales BM, Cain CJ, et al. NF-κB/MAPk activation underlie ACVR1-mediated inflammation in human heterotopic ossification. JCI Insight. 2018;3(22)e122958:1–21. https://doi.org/10.1172/jci.in....
23.
Haviv R, Moshe V, De Benedetti F, et al. Is fibrodysplasia ossificans progressiva an interleukin-1 driven autoinflammatory syndrome? Pediatric Rheumatology. 2019;17(84):1–7. https://doi.org/10.1186/s12969....
24.
Wolken DMA, Idone V, Hatsell SJ, et al. The obligatory role of Activin A in the formation of heterotopic bone in Fibrodysplasia Ossificans Progressiva. Bone. 2018;109:210–217. https://doi.org/10.1016/j.bone....
25.
Anthonissen J, Steffen CT, Hofmann A, et al. The pathogenesis of heterotopic ossification after traumatic brain injury. A review of current literature. Acta Orthop. Belg. 2020;86(3):369–377.
26.
Vincent S, Marty L, Fort P. S26 ribosomal protein RNA: an invariant control for gene regulation experiments in eucaryotic cells and tissues. Nucleic Acids Research. 1993;21(6):1498. https://doi.org/10.1093/nar/21....
27.
Themo Fisher Scientyfic. SYBR® Green PCR Master Mix and SYBR® Green RT- PCR Reagents Kit. www.assets.thermofisher.com/TFS-Assets/LSG/manuals/cms_041053.pdf (acesso: 05.01.2022).
28.
Livak KJ, Schmittgen TD. Analysis of relative gene expression data using real- time quantitative PCR and the 2-ΔΔCT method. Methods. 2001;25(4):402–408. https://doi.org/10.1006/meth.2....
29.
Kaplan FS, Al Mukaddam M, Baujat G, et al. The Medical Management Of Fibrodysplasia Ossificans Progressiva: Current Treatment Considerations. Proc Intl Clin Council FOP. www.fopaustralia.org/ wp-content/uploads/2019/06/FOP_TREATMENT_GUIDELINES_June_2019.pdf (acesso: 12.09.2023).
30.
Theda C, Hwang SH, Czajko A, et al. Quantitation of the cellular content of saliva and buccal swab samples. Scientific reports. 2018;8(6944):1–8. https://doi.org/10.1038/s41598....
31.
Shaikh U, Khan A, Kumari P, et al. Novel Therapeutic Targets for Fibrodysplasia Ossificans Progressiva: Emerging Strategies and Future Directions. Cureus. 2023;15(7) e42614:1–9. https://doi.org/10.7759/ cureus.42614.
32.
Dongen JV, Ehli EA, Jansen R, et al. Genome-wide analysis of DNA methylation in buccal cells: a study of monozygotic twins and mQTLs. Epigenetics & Chromatin. 2018;11(54):1–14. https://doi.org/10.1186/ s13072-018-0225-x.
33.
Khurshid Z, Zafar MS, Khan RS, et al. Role of Salivary Biomarkers in Oral Cancer Detection. Adv Clin Química. 2018;86:23–70. https://doi. org/10.1016/bs.acc.2018.05.002.
34.
Kumar P, Gupta S, Das BC. Saliva as a potential non-invasive liquid biopsy for early and easy diagnosis/prognosis of head and neck câncer. Translational Oncology. 2024;40(101827):1–17. https://doi.org/10.1016/j. tranon.2023.101827.
35.
Ferrari H, Pezzi ME, Cassi D, et al. Salivary Cytokines as Biomarkers for Oral Squamous Cell Carcinoma: A Systematic Review. Int J Mol Sci.2021;22(6795):1–14. https://doi.org/10.3390/ijms22....
36.
Joesch-Cohen LM, Glusman G. Differences between the genomes of lymphoblastoid cell lines and blood-derived samples. Advances in Genomics and Genetics. 2017;2017(7):1–9. https://doi.org/10.2147/AGG. S128824.
37.
Omi N, Tokuda Y, Ikeda Y, et al. Efficient and reliable establishment of lymphoblastoid cell lines by Epstein-Barr virus transformation from a limited amount of peripheral blood. Scientific Reports. 2017;7(43833):1– 10. https://doi.org/10.1038/srep43....
38.
Lopez-Lozano AP, Arevalo-Niño K, Gutierrez-Puente Y, et al. SSEA-4 positive dental pulp stem cells from deciduous teeth and their induction to neural precursor cells. Head Face Med. 2022;18(9):1–11. https://doi. org/10.1186/s13005-022-00313-6.
39.
Hino K, Horigome K, Nishio M, et al. Activin-A enhances mTOR signaling to promote aberrant chondrogenesis in fibrodysplasia ossificans progressive. J Clin Invest. 2017;127(9):3339–3352. https:// doi.org/10.1172/JCI93521.
40.
Hsieh HHS, Chung MT, Allen RM, et al. Evaluation of Salivary Cytokines for Diagnosis of both Trauma-Induced and Genetic Heterotopic Ossification. Frontiers in Endocrinology. 2017;8(74):1–8. https://doi.org/10.3389/fendo.....
We process personal data collected when visiting the website. The function of obtaining information about users and their behavior is carried out by voluntarily entered information in forms and saving cookies in end devices. Data, including cookies, are used to provide services, improve the user experience and to analyze the traffic in accordance with the Privacy policy. Data are also collected and processed by Google Analytics tool (more).
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.