Online first
About the Journal
Current issue
Archive
Publication Ethics
Anti-Plagiarism system
Instructions for Authors
Instructions for Reviewers
Editorial Office
Editorial Board
Contact
Reviewers
All Reviewers
2025
2024
2023
2022
2021
2020
2019
2018
2017
2016
General Data Protection Regulation (RODO)
RESEARCH PAPER
Seroprevalence of Bordetella pertussis antibodies among a group of young adults
1
Students Scientific Association at the Chair and Department of Medical Microbiology, Medical University, Lublin, Poland
2
Chair and Department of Medical Microbiology, Medical University of Lublin, Lublin, Poland
Corresponding author
J Pre Clin Clin Res. 2021;15(3):126-128
KEYWORDS
TOPICS
ABSTRACT
Introduction and objective:
Bordetella pertussis is a Gram-negative rod which attacks the respiratory system and causes whooping cough disease. Despite the high percentage of childhood vaccination amongst the population an increasing number of pertussis cases have been observed over time, especially among adolescents and young adults.
Objective:
The aim of the study was to analyze the concentration of IgG anti-PT Bordetella antibodies in the serum of young adults in the aspect of maintaining immunity.
Material and methods:
The study group consisted of 30 patients, medical students aged 21–26 years. They were interviewed with a questionnaire, which asked about their general health, medications taken, basic information about whooping cough, or vaccination status against B. pertussis. The anti-PT Bordetella levels were measured in the collected serum by a commercial ELISA kit (enzyme-linked immunosorbent assay).
Results:
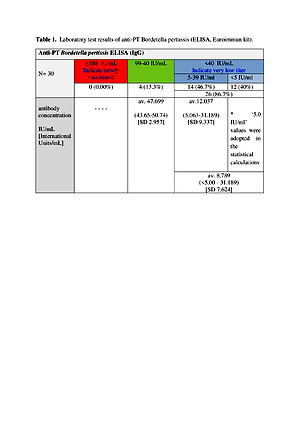
All patients had previously received a mandatory vaccination for pertussis as a child, and 36.6%had received a booster dose after the age of 18. The total average antibody level was 13.977 IU/mL (range: <5.00 – 50.74) and 40% of samples had a very low concentration (<5 IU/mL).
Conclusions:
Current findings suggest that post-vaccine immunity decreases over time, while infection susceptibility among younger age groups who have not yet been fully immunized has increased. Methods that could prevent and enable risk stratification of whooping cough include the screening test for anti-PT Bordetella antibody, the improvement of diagnostics, and an additional booster dose.
Bordetella pertussis is a Gram-negative rod which attacks the respiratory system and causes whooping cough disease. Despite the high percentage of childhood vaccination amongst the population an increasing number of pertussis cases have been observed over time, especially among adolescents and young adults.
Objective:
The aim of the study was to analyze the concentration of IgG anti-PT Bordetella antibodies in the serum of young adults in the aspect of maintaining immunity.
Material and methods:
The study group consisted of 30 patients, medical students aged 21–26 years. They were interviewed with a questionnaire, which asked about their general health, medications taken, basic information about whooping cough, or vaccination status against B. pertussis. The anti-PT Bordetella levels were measured in the collected serum by a commercial ELISA kit (enzyme-linked immunosorbent assay).
Results:
All patients had previously received a mandatory vaccination for pertussis as a child, and 36.6%had received a booster dose after the age of 18. The total average antibody level was 13.977 IU/mL (range: <5.00 – 50.74) and 40% of samples had a very low concentration (<5 IU/mL).
Conclusions:
Current findings suggest that post-vaccine immunity decreases over time, while infection susceptibility among younger age groups who have not yet been fully immunized has increased. Methods that could prevent and enable risk stratification of whooping cough include the screening test for anti-PT Bordetella antibody, the improvement of diagnostics, and an additional booster dose.
Skiba A, Raman S, Rzucidło I, Sereda D, Kozioł MM. Seroprevalence of Bordetella pertussis antibodies among a group of young adults. J Pre-
Clin Clin Res. 2021; 15(3): 126–128. doi: 10.26444/jpccr/141918
REFERENCES (12)
1.
Prygiel M, Mosiej E, Zasada AA. Przyczyny wzrostu zachorowalności na krztusiec. Postępy Mikrobiol. 2020; 59(1): 11–24.
2.
Libster R, Edwards KM. Re-emergence of pertussis: what are the solutions? Expert Rev. Vaccines 2012; 11(11): 1331–1346. doi: 10.1586/erv.12.118.
3.
Rumik A, Paradowska-Stankiewicz I, Rudowska J, et al. Krztusiec w Polsce w 2017 roku. Przegl Epidemiol. 2019; 73(6): 289–295.
4.
Nieves DJ, Heininger U. Bordetella pertussis. Microbiol. Spectrum 2016; 4(3): 274–75. doi: 10.1128/microbiolspec.EI10–0008–2015.
5.
Maryland Department of Health, “Local Health Department Guidelines for the Epidemiological Investigation and Control of Pertussis (Whooping Cough),” no. February, 2018, [Online]. Available: https://phpa.hea lt h.mar yland.gov/IDEHASharedDocuments/Per tussis Investigation Guidelines Revised February 2018 Final-ES.pdf.
6.
Ebell MH, Marchello C, Callahan M. Clinical diagnosis of Bordetella pertussis infection: Asystematic review. J Am Board Fam Med. 2017; 30(3): 308–319. doi: 10.3122/jabfm.2017.03.160330.
7.
Torzsa P, Devadiga R, Tafalla M. Seroprevalence of Bordetella pertussis antibodies in adults in Hungary: Results of an epidemiological cross-sectional study. BMC Infect Dis. 2017; 17(1): 242. doi: 10.1186/s12879–017–2356–2. PMID: 28376739.
8.
Jõgi P, Oona M, Toompere K, et al. Seroprevalence of IgG antibodies to pertussis toxin in children and adolescents in Estonia. Vaccine 2014; 32(41): 5311–5315. doi: 10.1016/j.vaccine.2014.07.066.
9.
Palazzo R, Carollo M, Fedele G, et al. Evidence of increased circulation of Bordetella pertussis in the italian adult population from seroprevalence data (2012–2013). J Med Microbiol. 2016; 65(7): 649–657. doi: 10.1099/jmm.0.000264.
10.
Pavlopoulou ID, Syriopoulou V, Daikos GL, et al. Pertusssis seroprevalence in different age groups in Greece. Scand J Infect Dis. 2007; 39(1): 14–18. doi: 10.1080/00365540600891299.
11.
Eberhardt CS, Siegrist CA. What Is Wrong with Pertussis Vaccine Immunity? Cold Spring Harb. Perspect Biol. 2017; 9(12): a029629. doi: 10.1101/cshperspect.a029629.
12.
Sompagdee N, Anuwutnavin S, Phongsamart W, et al. Seroprevalence of Bordetella pertussis antibodies and anti-pertussis antibody response after a single dose of reduced-antigen combined diphtheria, tetanus, and acellular pertussis vaccine (Tdap) in pregnant Thai women. Vaccine 2020; 38(12): 2725–2733. doi: 10.1016/j.vaccine.2020.01.074.
We process personal data collected when visiting the website. The function of obtaining information about users and their behavior is carried out by voluntarily entered information in forms and saving cookies in end devices. Data, including cookies, are used to provide services, improve the user experience and to analyze the traffic in accordance with the Privacy policy. Data are also collected and processed by Google Analytics tool (more).
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.