Online first
About the Journal
Current issue
Archive
Publication Ethics
Anti-Plagiarism system
Instructions for Authors
Instructions for Reviewers
Editorial Office
Editorial Board
Contact
Reviewers
All Reviewers
2025
2024
2023
2022
2021
2020
2019
2018
2017
2016
General Data Protection Regulation (RODO)
REVIEW PAPER
John William Trevan’s concept of Median Lethal Dose (LD50/LC50) – more misused than used
1
International Institute of Biotechnology and Toxicology (IIBAT), Padappai, India
2
Ex-Cabinet Secretary Researcher, Food Safety Commission of Japan, Tokyo, Japan
3
Trichinopoly, India
4
TOXINDIA, Maharashta, India
5
SASTRA Deemed University, Thanjavur, India
6
Sivavilas, India
Corresponding author
Sadasivan Kalathil Pillai
International Institute of Biotechnology and Toxicology (IIBAT), Padappai, Kancheepuram Dist, India
International Institute of Biotechnology and Toxicology (IIBAT), Padappai, Kancheepuram Dist, India
J Pre Clin Clin Res. 2021;15(3):137-141
KEYWORDS
TOPICS
ABSTRACT
Introduction:
Median lethal dose (MLD) has been a controversial subject among biologists and animal ethicists since its inception in 1927 by Trevan. Toxicologists use MLD (LD50) as the first step to assess the toxicity of a substance. Animal ethicists criticize LD50 tests because animals suffer pain, and LD50 is irreproducible. The disadvantage of classifying chemicals based on LD50, the importance of the ‘characteristics’ proposed by Trevan, and the ideal mortality range for determining the best estimate of LD50 are also discussed.
Objective:
The aim of this review was to understand Trevan’s concept of LD50 and the method of Litchfield and Wilcoxon (L and W), and Finney’s probit analysis for LD50 determination
Materials and method:
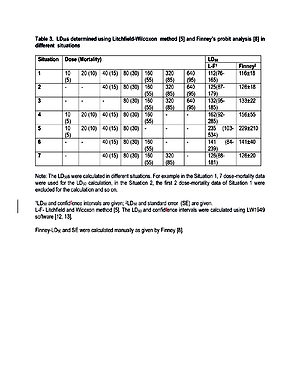
A literature survey was conducted using Google search and Pubmed. Simulated data set was used for identifying the ideal mortality range for calculating the ‘best estimate’ of LD50.
Brief description of the state of knowledge:
After Trevan, the extensively used classical methods for LD50 determination are Finney’s probit analysis and the L and W method. Animal ethicists questioned LD50, because of its irreproducibility. Presently used methods for LD50 tests do not provide information on the dose-response, hence assessing the complete spectrum of toxicity is not possible. However, LD50 is used to classify chemicals.
Conclusions:
'The 'characteristic' is more specific than the slope or LD50 of a dose-response curve. LD50 does not manifest the exact nature of the toxicity of a substance; hence, classifying chemicals based on LD50s may have little relevance
Median lethal dose (MLD) has been a controversial subject among biologists and animal ethicists since its inception in 1927 by Trevan. Toxicologists use MLD (LD50) as the first step to assess the toxicity of a substance. Animal ethicists criticize LD50 tests because animals suffer pain, and LD50 is irreproducible. The disadvantage of classifying chemicals based on LD50, the importance of the ‘characteristics’ proposed by Trevan, and the ideal mortality range for determining the best estimate of LD50 are also discussed.
Objective:
The aim of this review was to understand Trevan’s concept of LD50 and the method of Litchfield and Wilcoxon (L and W), and Finney’s probit analysis for LD50 determination
Materials and method:
A literature survey was conducted using Google search and Pubmed. Simulated data set was used for identifying the ideal mortality range for calculating the ‘best estimate’ of LD50.
Brief description of the state of knowledge:
After Trevan, the extensively used classical methods for LD50 determination are Finney’s probit analysis and the L and W method. Animal ethicists questioned LD50, because of its irreproducibility. Presently used methods for LD50 tests do not provide information on the dose-response, hence assessing the complete spectrum of toxicity is not possible. However, LD50 is used to classify chemicals.
Conclusions:
'The 'characteristic' is more specific than the slope or LD50 of a dose-response curve. LD50 does not manifest the exact nature of the toxicity of a substance; hence, classifying chemicals based on LD50s may have little relevance
Sadasivan KP, Katsumi K, Mathews M, Thomson M, Bhavana S, Parvathy S. John WilliamTrevan’s Concept on Median Lethal Dose (LD50/LC50)-
More Misused than Used. J Pre-Clin clin Res. 2021; 15(3): 137–141. doi: 10.26444/jpccr/139588
REFERENCES (27)
1.
Trevan JW. The error of determination of toxicity. Proc R Soc Lond. 1927; B.101: 483–514.
2.
Gad SC. Rodents model for toxicity testing and biomarkers. In: Gupta RC, editor. Biomarkers in toxicology. Academic Press; 2014. p. 7–69.
3.
Agrawal SP, Saxena VL. Prediction of LD50 of some common pesticides through QSAR (an alternative method to save experimental animals). Int J Pharm Sci Res. 2014; 5(12): 5356–5373. doi: 10.13040/IJPSR.0975-8232.5 (12). 535 6 -73.
4.
Badanthadka M, Mehendale HM. Hexachlorocyclopentadiene. In: Wexler P, editor. Encyclopedia of Toxicology. 33rd ed. Academic Press; 2014. p. 877–881.
5.
Fuentes D, Ray SD, Holstege CP. Anxiolytics, In: Wexler P, editor. Encyclopedia of Toxicology. 3rd ed. Academic Press; 2014. p. 280–286.
6.
Erhirhie EO, Ihekwereme, CP, Ilodigwe EE. Advances in acute toxicity testing: strengths, weaknesses and regulatory acceptance. Interdiscip Toxicol. 2018; 11: 5–12.
7.
Litchfield JT Jr, Wilcoxon F. A simplified method of evaluating dose-effect experiments. J Pharmacol Exp Ther. 1949; 96(2): 99–113.
9.
Finney DJ. Probit Analysis. A statistical treatment of sigmoid response curve. 2nd ed. Cmabridge; 1952.
10.
Tallarida RJ, Murray RB. Litchfield and Wilcoxon: Confidence Limits of ED50. In: Manual of Pharmacologic Calculations. New York: Springer; 1987. https://doi.org/10.1007/978-1-....
11.
Adams JV. Introduction to LW1949; 2017. https://cran.r-project.org/web... vignettes/ Intro.html#:~:text=The%20R%20package%20LW1949%20automates,the%20chi%2Dsquared%20statistic
12.
Adams JV, Slaght KS, Boogaard MA. An automated approach to Litchfield and Wilcoxon’s evaluation of dose-effect experiments using the R package LW1949. Environ Toxicol Chem. 2016; 35(12): 3058–3061. doi: 10.1002/etc.3490.
13.
Dearden JC, Hewitt M. Prediction of human lethal doses and concentrations of MEIC chemicals from rodent LD50 values: An attempt to make some reparation. Altern Lab Anim. 2021; 49(1–2): 10–21. doi: 10.1177/0261192921994754.
14.
Betz JM, Brown PN, Roman MC. Accuracy, precision, and reliability of chemical measurements in natural products research. Fitoterapia. 2011; 82(1): 44–52. doi: 10.1016/j.fitote.2010.09.011.
16.
Zhang H, Chen W, Sun K. Mendelism: New insights from Gregor Mendel’s lectures in Brno. Genetics. 2017; 207(1): 1–8. doi: 10.1534/genetics.117.201434.
17.
van Dijk PJ, Weissing FJ, Ellis THN. How Mendel’s interest in inheritance grew out of plant improvement. Genetics. 2018; 210(2): 347–355. doi: 10.1534/genetics.118.300916.
18.
Boyd EM. Predictive Toxicometricsm Scientechnica (Publishers) Ltd., Bristol, 1972.
19.
OECD. Test No. 408: Repeated Dose 90-Day Oral Toxicity Study in Rodents, OECD Guidelines for the Testing of Chemicals, Section 4, OECD Publishing, Paris, 2018. https://doi.org/10.1787/978926....
20.
Kobayashi K, Pillai KS. A Handbook of Applied Statistics in Pharmacology. New York: CRC Press; 2013.
21.
Jen-Yin G, Richard JW, Libby D, et al.. Development and use of in vitro alternatives to animal testing by the pharmaceutical industry 1980–2013. Toxicol Res. 2015; 4: 1297–1307.
22.
Sonali K, Doke S, Dhawale C. Alternatives to animal testing: A review. Saudi Pharm J. 2015; 23: 223–229.
23.
Tsatsakis AM, Vassilopoulou L, Kovatsi L, et al. The dose response principle from philosophy to modern toxicology: The impact of ancient philosophy and medicine in modern toxicology science. Toxicol Rep. 2018; 5: 1107–1113. doi: 10.1016/j.toxrep.2018.10.001. PMID: 30450285; PMCID: PMC6226566.
24.
Lipnick RL, Cotruvo JA, Hill RN, et al. Comparison of the up-and-down, conventional LD50, and fixed-dose acute toxicity procedures. Food Chem Toxicol. 1995; 33(3): 223–231. doi: 10.1016/0278-6915(94)00136-c. PMID: 7896233.
25.
Barker-Treasure C, Coll K, Belot N, et al. Non-animal replacements for acute toxicity testing. Altern Lab Anim. 2015; 43(3): 199–203. doi: 10.1177/026119291504300308. PMID: 26256397.
26.
Gadaleta, D, Vuković, K, Toma, C, et al. SAR and QSAR modeling of a large collection of LD50 rat acute oral toxicity data. J Cheminform. 2019; 11(18): 1–16. https://doi.org /10.1186/s13321- 019- 0383-2.
27.
Rang HP, Hill RG. Assessing drug safety, in Rang HP, Hill RG editors, Drug discovery and development. 2nd ed. Churchill Livingstone; 2013. p. 211–225.
We process personal data collected when visiting the website. The function of obtaining information about users and their behavior is carried out by voluntarily entered information in forms and saving cookies in end devices. Data, including cookies, are used to provide services, improve the user experience and to analyze the traffic in accordance with the Privacy policy. Data are also collected and processed by Google Analytics tool (more).
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.